If you want to know why real-time monitoring and tracking of the cold pharmaceutical chain is important, read this article.
Why is Pharmaceutical Cold Chain Real-Time Monitoring and Tracking Important?

To ensure safety and product quality during pharmaceutical distribution, the products must be stored properly and at the right temperature.
All pharmaceutical products, including vaccines, pills, and blood products, are sensitive to temperature and humidity, and not handling the distribution of those products properly can completely damage the product or reduce its efficiency.
Environmental Control of Pharmaceutical Products
The pharmaceutical industry is among the most profitable, with an estimated worth of $1170 billion in 2021. The constant growth of the industry implies strict regulations regarding handling, storing, and transportation. To ensure the proper handling of sensitive pharmaceutical products, the World Health Organization has published guidelines that every pharmaceutical company needs to follow.
Here are some of the guidelines that pharmaceutical companies are obligated to follow.
Importation
Properly equipped pots of entries must import temperature and time-sensitive pharmaceuticals. After the medications are offloaded, the transport to the pharmaceutical company should be minimized, and the pharmaceutical products must be carried in temperature-controlled storage. This temporary storage has the best conditions for the pharmaceutical products while waiting for customs clearance.
Proper Warehouse
After the products pass the customs, they must be transferred to safe storage in a controlled environment. Pharmaceutical companies must carefully select the proper warehouse to minimize the damage that natural disasters can eventually cause. The warehouse must be designed to handle temperature and time-sensitive pharmaceutical products and be energy efficient. The pharmaceutical products must not be exposed to dirt, dust, rain, and direct sunlight during loading and delivery. A proper warehouse must also have a quarantine area in case there’s an accident with faulty pharmaceutical products.
Security
Pharmaceutical products are a target for robbers because they have a high value. Some pharmaceutical products contain restricted substances that need secure storage, so the pharmaceutical company needs to take extra security measures.
Additional Power Supply
Pharmaceutical companies must have a backup power system. Sometimes the power can go down, and that can cause the chillers and freezers to stop working and cause the pharmaceutical products to go bad. A backup power system or generator will ensure that there’s electricity even if the power is off.
Temperature Control
Several rules must be followed to ensure that temperature-sensitive pharmaceutical products are handled properly. The freezer rooms and temperature-controlled storage must:
- Have proper equipment for temperature monitoring
- Have proper equipment for temperature monitoring
- Have accurate sensor monitoring for humidity
- Have an alarm that will alert when there’s something wrong with the temperature
- Have sensors that are accurate to +/- 0.5 °C
- Have an excellent control system
Cold Chain Transport of Temperature Sensitive Pharmaceutical Products
A cold chain is a temperature-controlled supply chain that involves constant refrigeration of pharmaceutical products. A cold chain transport is important because it preserves the usability and quality of pharmaceutical products. The pharmaceutical industry transports a variety of products every day, such as:
- Over-the-counter medicines
- Biologics such as blood, vaccines, and tissue
- Prescribed medications
- Raw pharmaceutical materials
To preserve the efficacy of the pharmaceutical products, the products must be frozen or chilled. Products that need to be transported frozen must be in a temperature colder than -15 °C. Commonly, frozen products undergo a laboratory check-up before being used because it can be difficult to see whether the product has been re-frozen, etc. Frozen cargo is mainly used for organs and tissues.
Products that need a chilled environment must be transported to a temperature of 2 °C-8 °C. These products must not be frozen or exposed to a higher temperature because it will damage the efficiency of the product.

What Can Happen if the Pharmaceutical Products Are Not Transported at The Right Temperature?
The consequences can be life-threatening when temperature-sensitive pharmaceutical products are not handled properly during transport. Products that are sensitive to high temperatures can deteriorate if they’re exposed to a higher temperature. The active ingredients content will decrease, possibly toxic the product’s properties. When exposed to high temperatures, some products can depreciate their formulation properties.
Some pharmaceutical products are sensitive to cold temperatures, and when they’re exposed, they can lose their therapeutic properties. Some special pharmaceutical products can even harm people’s health if they’re not transported and kept at the right temperature.
Other pharmaceutical products, like vaccines, require a temperature from 2 °C up to 8°C. Exposure of vaccines to temperatures below and higher and exposure to light can cause the vaccine to lose its efficiency and potency.
Cold-supply chain is extremely important to avoid unnecessary deterioration of pharmaceutical products. If the temperature, humidity, and other conditions are properly monitored during transport, there will be no room left for mistakes, and not one pharmaceutical product will be damaged.

How Can UbiBot Help?
UbiBot GS1 is a device that can improve the cold-chain pharmaceutical supply because it can effectively regulate the transportation of drugs, including holding tanks and refrigerated trucks.
GS1 is an industrial-grade cloud-based wireless data logger monitoring temperature, humidity, and ambient light. The device works on Wi-Fi and has two cellular types: connection and GPS location tracking.
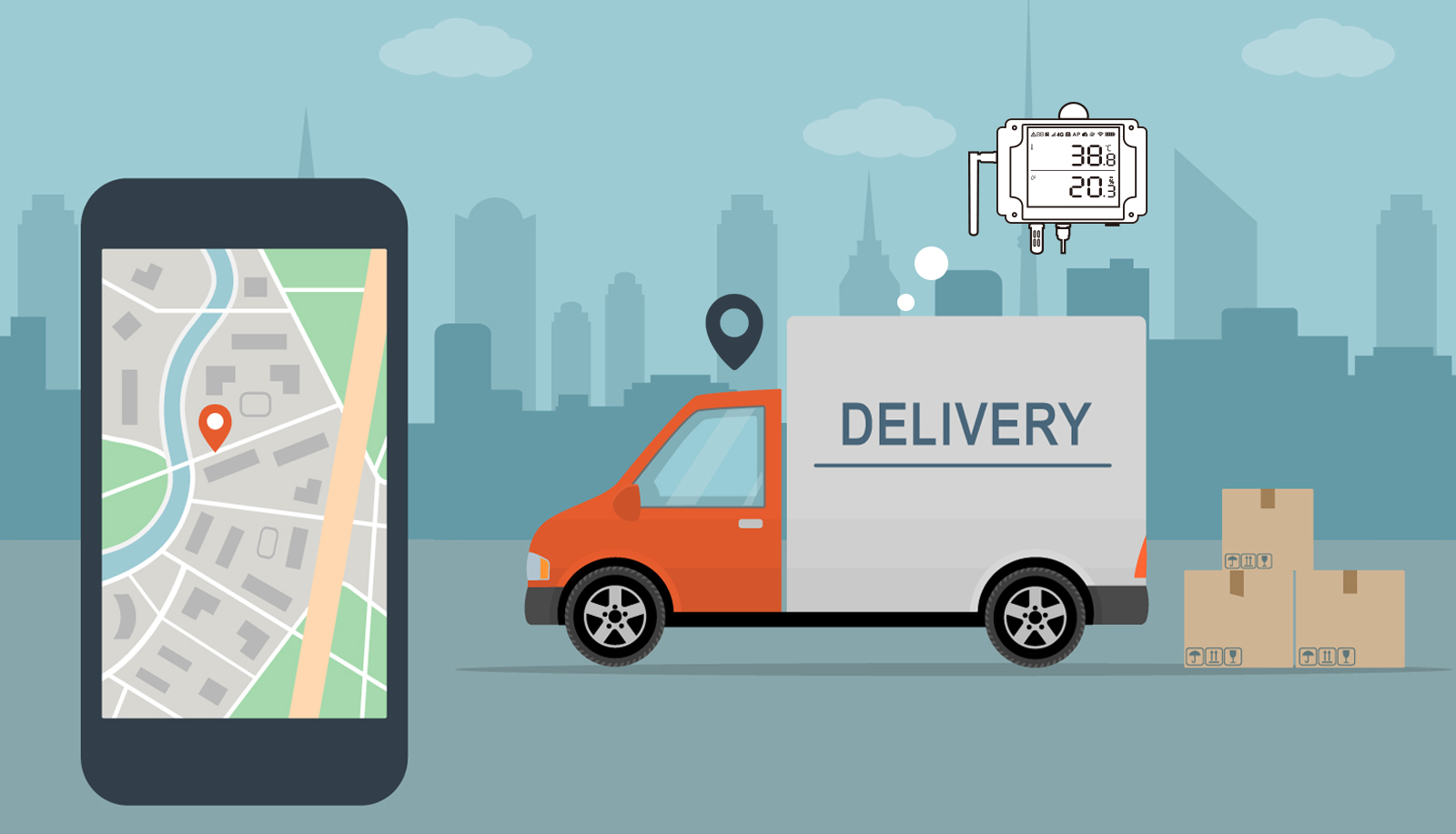
The special thing about GS1 is that it provides real-time detection during the pharmaceutical product’s transportation and enables pharmaceutical companies to trace the entire logistics fully.




